Written by Joe Ballenger
Hi, I’m on a Lego League team with my school and we are wanting to know more about spiders for our research project. Some questions I hope you can help us with are, what makes a spiders web so strong and sticky? Is it even stronger if it were bigger to hold a person?
Thank you from my whole team!!
Spider silk is really strong stuff. A single strand of spider silk can instantly catch and stop a flying insect tens of thousands of times its weight, without breaking. Scientists are interested in harnessing this property for everyday wear, bulletproof vests, and other protective clothing. There’s a lot of interest in how to do this, from a chemical standpoint.
So it’s a really neat question, and the Lego League is certainly interested in this from a structural chemistry perspective. However, there’s also a cultural component to this particular question I think is neat.
I hang out (pun intended) on ropes quite a bit, and I recently joined a gym which teaches a type of performance art called aerial silk. It’s a type of performance art where a dancer moves by suspending themselves by wrapping themselves in a strong fabric.
When starting to do research on this topic, I asked about the fabric used in this sort of performance. I was shocked to find that the fabric these people are using was nylon, and not silk.
So what is silk, why is it super strong, and why don’t we make climbing equipment with it?
So what is this stuff?
The first thing we should point out is that spider silk isn’t the strongest material known to man. Carbon-fiber can handle four times the load of spider silk, but it isn’t that elastic. As soon as carbon fiber stretches, even a small amount, it snaps. For that reason, it’s a good replacement for things that would normally be made out of metal…but it’s not a great replacement for fibers used in clothing. Steel has the same thing going on…its tensile strength is on par with spider silk, but it’s far heavier and not nearly as flexible.

A spider on a web eating shows us how many different types of silk spiders produce. A single web consists of a sturdy silk (dragline silk), connected to a stretchy silk (flagelliform silk), which is coated in a sticky substance (aggregate silk). The dragline and flagelliform silk are glued together with attachment cement (piriform silk), which is a fourth type of silk made from the silk glands. When the spider captures prey, it restrains its movement using aciniform silk. Not shown is the silk which coats eggs (cylindrical silk), or reinforcement silk (minor ampullate silk). Image credit: Jeroen Mul, via Flikr. License info: CC BY-NC-SA 2.0. Image modified from original.
The properties we like in spider silk isn’t its strength but its toughness. Although we may associate strength and toughness in things like character traits, they’re very different in the eyes of material scientists. While strength is how much weight you can dangle on a cable, toughness is how hard you can hit something without breaking it.
This brings us to spider silk, which again, is kind of a complicated topic because. There’s about 7 different types of silk spiders can spin, although many don’t have this arsenal at their disposal. These 7 types are used for a variety of purposes, have completely different properties, and different chemical compositions.
Sticky, Stretchy and Strong
A spider web is comprised mainly of three types of silk: Flagelliform, aggregate, and dragline. Dragline silk is the toughest silk, and is what scientists try to emulate when they try to make garments out of the stuff.
What makes spider silk so sticky?
Flagelliform silk is what captures the prey. It’s able to stretch and deform, stretching nearly 30x its length, without breaking. Its purpose is to arrest the speed of the insect, with the actual stopping power provided by the dragline silk it’s connected to. The flagelliform silk doesn’t actually grab onto the insect, however. That job is done by the aggregate silk.
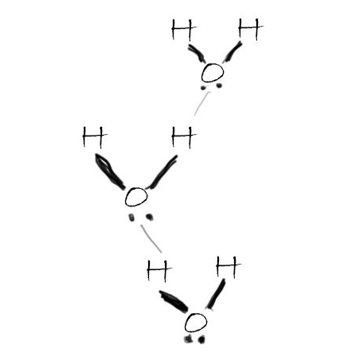
Hydrogen bonding demonstrated using water molecules. Individual hydrogen bonds are weak, but become strong when there are lots of them in a small area.
Aggregate silk is similar to silk in composition, except it has sugars stuck to the surface. These sugars are covered in OH groups, which are attracted to groups of atoms which have lots of electrons. These individual attractions are weak, but when there are a lot of them, they become very strong.
This trick is one which is repeated throughout the animal kingdom. Fish eggs, for example, use glycoproteins to stick to stuff. Urea, secreted in urine, is also efficient at hydrogen bonding. Not only is it used as glue to stick plywood together, glowworm fly larvae use it to make their silk sticky.
The strength of a spiderweb as a whole is due to its ability to stick to prey items, as well as its ability to slow them down without breaking.
Why is silk so strong?
Dragline silk is the easiest to harvest from spiders, and it’s also the toughest. Because it’s so easy to harvest, it’s the silk we know the most about.
Silk is a giant prion, a type of protein crystal. Inside the spinerette, it’s comprised of bits of silk that are suspended in a liquid medium. As it’s extruded, the molecules link up and create a giant thread which comes out of the spider’s bum.
How they link up is a matter of debate, and I’m not going to get into it here. However, thanks to X-ray crystallography, we know what the molecules look like and it will be a lot simpler to start at the molecule and work our way up.
From molecules to spiderwebs
Since this question came from a team which plays with legos competitively, let’s take a look at how these molecules are linked together. The important structures in the molecule are stiff parts of the protein called beta-sheets, which reinforce themselves through hydrogen bonds:
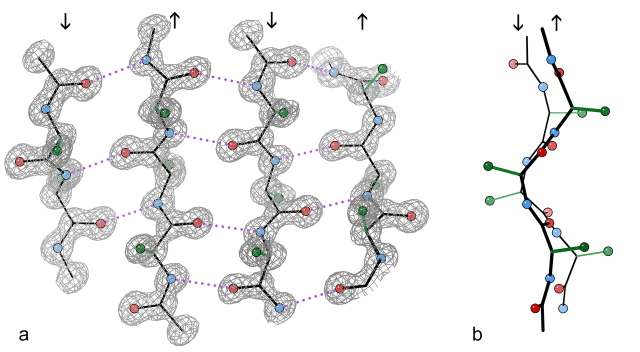
How beta-sheets work. The figure on the left is what we’re interested in. Dotted lines are hydrogen bonds between protein segments. Image credit: Dcrjsr, via Wikipedia commons. Image credit: CC-by-3.0
Here is an X-ray crystallography ‘photograph’ of an individual silk molecule:

Spider silk under unloaded (initial), loaded (extension), and breaking (failure) stress. Image credit: Brahtzel & Buhler 2011
Those yellow segments are beta-sheets, and they keep the protein together. The stuff at the end is mostly made up of another type of structure, called alpha-helices. Beta-sheets keep the molecule together, while the alpha-helices allow the molecule to stretch a bit. Spider silk is a bunch of these molecules stuck together, and the way they’re stuck together reinforces each other by maximizing those hydrogen bonds.
Here is how those molecules are stuck together:
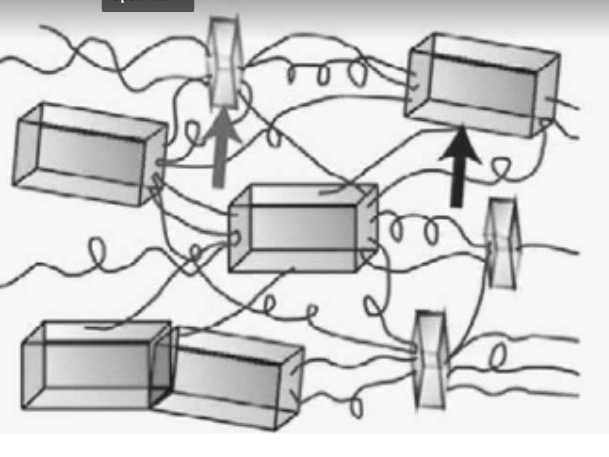
Image credit: Blackledge, 2012
Within the structure of spider silk, those individual molecules link up together to form crystals within the thread. Those crystals are loosely held together by more flexible strings. The hydrogen bonding within the beta-sheets creates a strong structure, which is reinforced at multiple levels. The flexible protein structures allows the spider silk to be flexible and stretchy.
This loose collection of crystals and protein structures forms a rope…and the spider spins multiple ropes. These ropes are held together with a couple of protein layers, and this provides another layer of reinforcement:
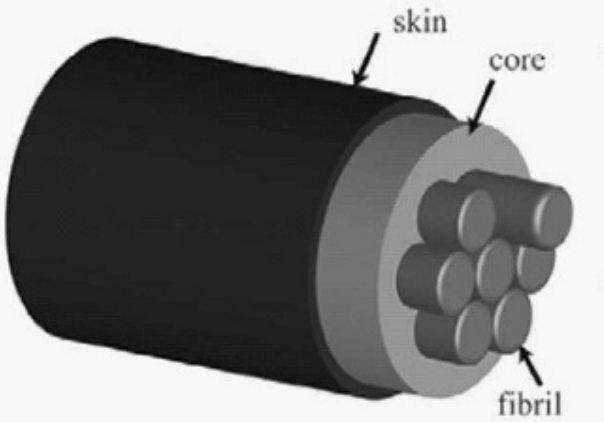
Image credit: Blackledge 2012
If scaled up, could we use silk to hold people?
This question is difficult to answer, because we can’t actually make big ropes out of spider silk yet. In order to be spun at the optimal strength, the chemical conditions of any silk produced outside of the animal’s body have to be perfect. We’re not quite at large-scale production yet, although we may be getting closer.
Regardless, it helps to have a comparison of some sort. Climbing ropes are made out of stretchy nylon, and a strand the strongest spider silk on record is 18 times stronger in proportion. Silkworm silk, which is produced in mass quantity, is about 6 times stronger in proportion. We’ve been mass-producing caterpillar silk for thousands of years, so this is a pretty good model.
It should be mentioned that the above information is comparing individually spun threads with a commercially prepared climbing rope. Preparation methods, humidity, the way the fabric is woven, and even things like dyes can effect the strength of commercially prepared products. While an individual strand of silk, either spider or caterpillar, is about as strong as an individual strand of nylon used in climbing rope…I’m not particularly happy with this comparison.
I feel like the only way properly answer this question is by looking at commercially prepared products designed to hold people. This is where aerial silk comes back in.
This is a dance performed on nylon fabric, which means I can compare the breaking strength of this fabric to commercially prepared silk fabric. Aerial silk fabric (again, made of nylon) can hold a little over 1100 kg. Surah, on the other hand, has a breaking strength of 30 kg. This is well below anything I’d ever support myself with. Ropes or aerial silks need to be able to hold at least 10 times your weight in order to be safely used.
There are applications of silk which have been used to hold people, but shortages and trade agreements after WWII changed silk from a fabric used to make these sorts of protective equipment to clothing. The way silk is prepared can drastically change its strength, so things made of silk may not be stronger than things made of nylon even though the individual threads may be stronger.
The Bottom Line

Spider silk (MA and Flag) compared to properties of different materials. Spider silk is extremely light and tough, although it is not as strong as other materials. Image Credit: Romer & Scheibel, 2008
The field of biomimetics has made some really big strides in reproducing spider silk in the past few years, but the vast differences in biochemistry between the spider’s spinerette and in-vitro production systems has made this difficult. The reputation of silk as a luxury good, and the associated economics, have favored nylon for use in safety equipment over silk despite the fact that silk is hypothetically better by many measurements.
Silk, whether from a spider or an insect, is a very strong material although this is often conflated with toughness. Your typical web-building spider dragline is actually about as strong as a typical nylon thread, although its toughness is more than twice as high. Commercial preparation can drastically change this for a number of reasons, and the same fiber at a different scale may have different properties because of a whole host of factors.
The biggest stride in making artificial spider silk happened this week. A group of scientists identified different sections of silk proteins which had better solubility under the conditions they were using to make the silk, and fused them together. The result was a mass-producible spider silk that is half as tough and 1/4th as strong as the silk that spiders spin.
So we’re getting there…but we’ve got a long ways to go.
Works Cited
- Andersson, M., Jia, Q., Abella, A., Lee, X. Y., Landreh, M., Purhonen, P., … & Plaza, G. R. (2017). Biomimetic spinning of artificial spider silk from a chimeric minispidroin. Nature Chemical Biology.
- Bratzel, G., & Buehler, M. J. (2012). Molecular mechanics of silk nanostructures under varied mechanical loading. Biopolymers, 97(6), 408-417.
- Blackledge, T. A. (2012). Spider silk: a brief review and prospectus on research linking biomechanics and ecology in draglines and orb webs. Journal of Arachnology, 40(1), 1-12.
- Brown, C. P., Whaite, A. D., MacLeod, J. M., Macdonald, J., & Rosei, F. (2015). With great structure comes great functionality: Understanding and emulating spider silk. Journal of Materials Research, 30(01), 108-120.
- Hu, X., Vasanthavada, K., Kohler, K., McNary, S., Moore, A. M. F., & Vierra, C. A. (2006). Molecular mechanisms of spider silk. Cellular and Molecular Life Sciences CMLS, 63(17), 1986-1999.
- Römer, L., & Scheibel, T. (2008). The elaborate structure of spider silk. Prion.
- Tokareva, O., Jacobsen, M., Buehler, M., Wong, J., & Kaplan, D. L. (2014). Structure–function–property–design interplay in biopolymers: Spider silk. Acta biomaterialia, 10(4), 1612-1626.
Special thanks to J. Kim for their assistance in finding us some of the literature used for this post!


Pingback: Spider Silk and Hearing Aids – Anaheim & Brea Hearing Center
Pingback: Spiders and Webs | Westside Excellence in Youth