Written by Joe Ballenger
Here’s where we get to the meat of the question our follower asked…why use the antibiotic tetracycline as a switch? Why not something else?
Last week, we discussed a system which could potentially work for this sort of application by forcing mosquitoes to incorporate artificial building blocks into their biology. However, no research group has attempted to develop mosquitoes using Genetic Recoding technology.
@BugQuestions why use antibiotic? bc its understood? why not use a dietary nutrient mosq can’t easily acquire in nature?
— tmigratorius (@tmigratorius) February 6, 2016
To answer this, we need to look really deep into the biology of the mosquitoes Oxitec is using for RIDL. Specifically, we need to look at how insects are reared, how transgenic insects are created, and how different genetic switch systems work.
How are these mosquitoes made?
It’s at this point where I have to admit that there are parts of this I’m not familiar with, because I’ve never worked with transgenic insects. I know how the process is done in fruit flies, but I’ve never worked with mosquitoes. The process is the same, but some details might differ.
To make transgenic insects, you inject DNA into the eggs while the embryo is developing. The DNA needs to insert itself into cells which will eventually become the reproductive organs, so you can breed the gene into a laboratory population. The process is not very efficient, as relatively few insects will uptake the genes in this manner. In Drosophila, the standard model organism, the success rate is 3% or less. If you need the gene in a specific place, it will take a lot more tries since the genes land in random places. The video below shows how scientists inject DNA into fruit flies for this process.
Here’s where we can answer the first part of our follower’s question. We can’t use Genetic Recoding, because mosquitoes are hard to rear in captivity. With bacteria, you can insert genes into millions of cells with the effort it takes to insert genes into one mosquito. Culturing bacteria is easy, because their generation times are measured in hours. The generation time of mosquitoes is measured in weeks, and you’ll need a lot of mosquito generations to make Genomic Recoding work.
The technology for Genomic Recoding just isn’t advanced enough for this sort of use, sadly.
Why Tetracycline?
In order to use this sort of system, you need something you can turn on in the lab and off in the field. The antibiotic tetracycline is broken down in animals slowly, so it works pretty well as a switch. This sort of system was popular with medical researchers when these mosquitoes were developed, and is still in use in labs all around the world today. In fact, a lot of papers which describe transgenic mosquitoes developed for this sort of application do use this exact switch.
I’m going to point out that, while the equipment used is involved with antibiotic resistance in E.coli, it’s not really an antibiotic resistance gene. The machine which E. coli uses to fight off tetracycline is a pump called TetA, which pumps tetracycline out of the cell. The parts of the gene used in the mosquitoes, described in detail here, simply tell the bacteria to make the pump that confers resistance. Without that pump, there’s no resistance…and it’s unlikely this equipment will transfer into another genome. Anastasia Bodnar discusses this further on Biology Fortified.
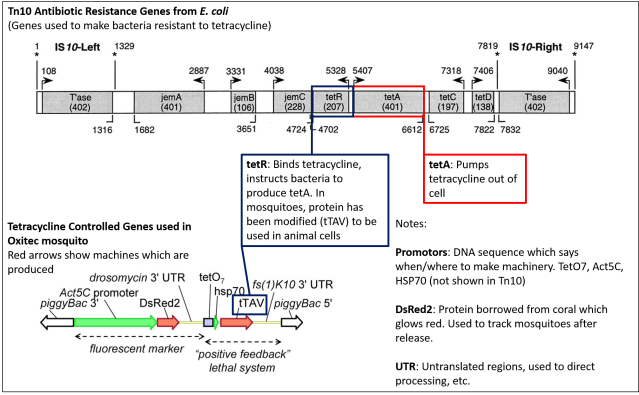
The portion of the tetracycline resistance gene which allows bacteria to survive a dose of tetracycline (tetA) has not been introduced to mosquitoes. The switch (tetR, modified to tTA) which tells bacteria to make that pump is what has been inserted. Figures from Chalmers et. al (2000) and Phuc et. al (2007) modified from originals by Joe Ballenger to show components.
By placing other stuff in place of that pump, you can get the mosquitoes to change their biology in predictable ways. For example, Oxitec has produced another transgenic mosquito strain where females don’t make wings outside the lab. Because you can switch parts in and out, there are a bunch of potential targets which could be used to re-enforce this system. So it’s very flexible.
This system is very well characterized, and is the most popular system for making these sorts of switches in whole animals. Consequently, a lot of people know how to use it. It’s not the only molecular switch on the market, and there are other sorts of switches that can be used.
What about other systems?
Here is a list of systems that I’m familiar with which could be used for switches. It’s not exhaustive by any means, but it’s a list of switch systems that I’ve either used or have considered using in the past.
- IPTG: Works great for bacteria, but animals have uptake issues. System has been occasionally adopted in mice, but not in insects as far as I know.
- Alcohol: Too toxic for use in mosquitoes, may be encountered in natural environment.
- Steroid induced: Works in cell culture, but mosquitoes regulate vital body functions by producing steroids. They’re always present in the mosquito, and turning off production would harm the insect. Using human steroids might effect development, as well.
- Heat-shock, light, etc: Works great in the lab, but natural situations might trigger some of these responses. Equipment linked to the switches can also happen at random times (e.g. they’re ‘leaky’). They’re too unreliable for this sort of application.
- CRISPR-CAS9: Only recently developed, but shows immense promise for these sorts of applications. Frankly, this is where I think the technology will go in the future.
By the way…I’m not familiar with every system on the market. If there’s one I missed, post a link in the comments and I’ll edit this section.
Why not target the disease-causing agent itself?
There are scientists who are working on this approach, and I think this approach does show promise, but I also think it’s hampered by a few things.
First, there’s not a lot of information about how these parasites interact with the mosquito. Mosquitoes try to fight off these diseases, but the parasites have their own counterattacks. The information needed to develop these approaches is rapidly advancing, but it’s still in its infancy at this point.
Second, and more importantly (in my opinion), there’s the question of economy. The same problems with vaccine development in people come up with mosquitoes, as well. Aedes can vector over 20 different viruses, each of which is unique. The viruses infect the mosquitoes in different ways, and something which targets one virus might not hit others.
Since we’re talking about Aedes, there are currently 4 different viral pandemics in need of control (Dengue, Yellow Fever, Chikungunya, Zika), plus a lot of smaller-scale potentially problematic viruses. Each modification may need to target a different virus unless scientists can find an important gene that’s basically the same between them. This approach might work for Malaria, but I’m not too sure it will work well for viruses.
There is an approach, using the symbiont Wolbachia, which appears to show promise in fighting Dengue because it creates a sort of broad-scale heightened immune system activation. Conceivably, this could also apply to other viruses…but it hasn’t been addressed in the literature. However, transgenic approaches tend to be a lot more narrow in scope so I’m not sure if this comparison applies here. I do think it’s worth mentioning, however.
While I think it’s a good idea worthy of further research, the TetOn/Off approach works in the here and now with what we have…and that’s really important. At this moment in time, targeting the mosquitoes is the easiest option and the furthest advanced.
Regulation
Regulation of these insects is an important topic, but unfortunately it’s one that I’ve got to skip over. Very briefly, because the technology is new, there are some things which still need to be worked through. Regulatory mechanisms are incredibly complex, and there’s just not enough room for an in-depth discussion because there are other things I want to cover.
PLOS Neglected Tropical Diseases has a series of papers (cited below) which give a good overview of these issues from the perspective of both industry and government regulators. They’re a few years old, and were published before the results of field trials were available, but they’re still relevant. I think they do a good job of describing the regulatory challenges, and I’ll refer you to those papers for details.
The regulation is based on existing protocols from agriculture (transgenic crops, biological control releases, and existing sterile insect programs), as well as drug trials. They’re regulated differently than GM crops, and many scientists consider these types of releases low-risk because they’re designed to extinguish themselves.
A lot of scientists demand that the data from lab trials be made publicly available through scientific publications, but this is hampered by the tendency of journals to only publish positive results. If there’s no difference in mating ability between a modified and unmodified mosquito, or if the mosquitoes don’t hurt stuff that eats them, it’s just hard to publish something like that. This is why PLOS ONE is such a great journal…they publish this stuff. So that information is out there now, and this problem should be less of a barrier as time goes on.
That’s not to say, however, that these releases are unregulated. The WHO has guidelines for this sort of testing. There’s also an international treaty, the Cartagena protocol, which regulates many aspects of transport of GM organisms. Regulation is mostly under the control of the countries the mosquitoes are released in, and details will be slightly different between countries. Right now, the mosquitoes have been approved for release in Brazil…but there’s some unknown issue holding them up.
The Bottom Line
The reason tetracycline is used for this sort of system is because it breaks down slowly in the mosquito, and won’t kill it. It’s also a system that’s very common and well understood. So there are good reasons tetracycline is the best choice, given the technology available to us.
I admit that I want to see this approach work, and I am by no means alone in the scientific community. Mosquito control isn’t very effective, and hasn’t changed much in 100 years. We need new tools. Badly.
One inevitable criticism of this series of articles is that I didn’t talk about the issues with regulation enough, and I accept that criticism. It’s something which should be tackled more thoroughly, but I want to direct my efforts towards discussing the results of the field trials. So that’s where we’re going to go next time. We’re going to travel out of the lab, and back to Brazil to see how mosquito control is working down there.
Further reading about transgenic mosquito regulation:
Works Cited:
Phuc, H. K., Andreasen, M. H., Burton, R. S., Vass, C., Epton, M. J., Pape, G., … & Coleman, P. G. (2007). Late-acting dominant lethal genetic systems and mosquito control. BMC biology, 5(1), 11.
Conflict of interest statement:
Joe Ballenger works as a contractor for a large biotechnology company, and develops new insect control components for use in agriculture. He has never worked for Oxitec, and has never worked with sterile insects. He has been writing about biotechnology long before he started his current position.
Special thanks to:
Karl Haro Von Mogel and Dasya Petranova for looking over the figure used in this post.
Jake Bova, for background information and general advice.


You worked so hard to write this up. You be awesome. Thank you!
LikeLike
A link to the Wolbachia efforts in Australia and SE Asia.
http://www.eliminatedengue.com/townsville
LikeLike
This blog post discusses a different technique (still in the laboratory stage, I understand), and it’s with malaria mosquitoes Anopheles gambiae.
>>So, the researchers genetically modified the enzyme so it would only be expressed during spermatogenesis, and it would have a really short half-life (it would break-down and become non-functional relatively quickly).
Translation: Male mosquitoes that had this gene engineered into their genomes would only make sperm with Y chromosomes. Very few or no X chromosomes. Meaning mostly all of their offspring would be male. The enzyme would be degraded by the time fertilization occurred, so it shouldnt accidentally slice up the necessary X-chromosome from mom-mosquito.
Lots and lots of viable baby male mosquitoes.<<
http://scienceblogs.com/erv/2014/06/11/gmo-mosquitoes-to-stop-mosquitoes/
Another article here:
http://www.theguardian.com/environment/2014/jun/10/gm-mosquitos-malaria-genetic-modification
LikeLike
And here is lab tested mechanism to genetically engineer the pathogen that causes malaria (Plasmodium falciparum), to act as a kind of vaccine … an immune response to non-pathogenic
>>The BioMed team’s plan is to intervene before the cycle can get started in the first place. Any incipient malaria infection has a brief window of vulnerability, right after a mosquito has delivered the parasite to the blood stream but before the parasite has taken refuge in red blood cells and begun to reproduce. The BioMed team would like that Plasmodium vanguard to find itself in a bloodstream already rife with antigens waiting to tag the parasite for termination.
The Plasmodium parasite is particularly difficult to work with, genetically, and this latest announcement represents a culmination of much genetic tinkering to figure out exactly which genes needed to be knocked out in Plasmodium flaciparum to render it innocuous yet inoculating. It took a “triple punch,” knocking out three key genes associated with pathogenicity, to render the parasite harmless. They tested their genetically attenuated parasite (GAP) in mice with human-equivalent red blood cells and livers. Even at high dosages, their modified parasite did not develop into an infection.<<
https://www.geneticliteracyproject.org/2015/01/04/malaria-vaccine-genetic-engineering-turns-parasite-into-vaccine-candidate/
LikeLike
…to non-pathogenic versions of the organism … [Edited add]
LikeLike
This one is still in the lab, and with Malaria mosquitoes (Anopheles gambiae) … only (mostly) males are produced, turning off the ability to produce X chromosomes.
>>So, the researchers genetically modified the enzyme so it would only be expressed during spermatogenesis, and it would have a really short half-life (it would break-down and become non-functional relatively quickly).
Translation: Male mosquitoes that had this gene engineered into their genomes would only make sperm with Y chromosomes. Very few or no X chromosomes. Meaning mostly all of their offspring would be male. The enzyme would be degraded by the time fertilization occurred, so it shouldnt accidentally slice up the necessary X-chromosome from mom-mosquito.
Lots and lots of viable baby male mosquitoes.<<
http://scienceblogs.com/erv/2014/06/11/gmo-mosquitoes-to-stop-mosquitoes/
Guardian article here:
http://www.theguardian.com/environment/2014/jun/10/gm-mosquitos-malaria-genetic-modification
LikeLike
More on gene edited mosquitoes using CRISPR-Cas9, as well as the Wolbachia alternative here:
http://blogs.plos.org/synbio/2016/02/16/controlling-the-transmission-of-zika-and-other-mosquitoe-borne-diseases-by-using-genetically-engineered-mosquitoes/
LikeLike