Written by Joe Ballenger
I was on Twitter recently, and one of my colleagues found a rather cool article by Luke Hollomon about plants and anesthesia.
When we’re working with insects in the lab, we often have to knock them out. There’s ethical reasons for this, but it’s also because we don’t want them moving. Either to prevent escape, or to keep them from thrashing when we perform surgery.
It’s not as important in the plant world, but anesthesia is potentially an important method of probing the biological responses in plants. I’m currently studying a type of plant movement, the motion of leaves when plants are exposed to other plants. I’ve toyed around with the idea of using anesthesia as a probe to determine what pathways are involved with this motion; an idea I got from my years of studying insect biology.
I’d definitely reccomend Holloman’s article, because it’s interesting and well written. However, the framing of some of these conversations centered around the article gave me a little bit of a cause for concern. There’s a lot of unknowns about how anesthesia works, but it’s not a complete biological mystery. We know enough about it to keep folks alive, what medications to avoid, and all of that.
While we need to acknowledge that no medical intervention is without risk, we understand anesthesia well enough to use it safely.
So this is a really interesting topic to explore: Why does anesthesia work on plants AND animals?
To answer this, we’re going to need to discuss why they work on bacteria.
Let’s Knock You Out For a Minute…
So, there are a lot of anesthetics. Depending on the specific chemical, you can block individual nerves, paralyze a patient, erase memories and straight-up knock them out. Each of these has a unique way of working, or Mode Of Action (MOA from here on out). Some anesthetics, like ketamine and scopalomine are well enough understood to point out which specific pathways are blocked.
The stuff we’re talking about today are decidedly not in that class.
Sending Out Signals
I could write this article about almost any inhalable anesthetic, but I think an exploration of isoflurane is a good candidate to describe why an anesthetic would work on just about any life form. It’s common, and it’s as simple as anything in this topic is gonna get.
So, the first thing to know is that in biology, there’s only so many ways to do something. Dolphins sort of look like fish because that’s a good way to move in a watery. Similarly, there’s only so many ways to send signals across a cell.
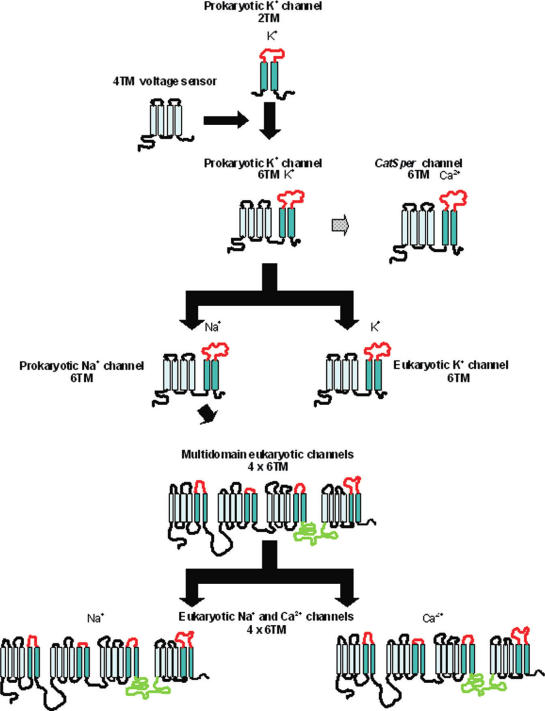
A lot of this stuff started early in evolutionary history. Bacteria need to be able to sense salt content of their environment. Too much, you shrivel up and die because your proteins can’t function. Too little, and you explode.
To solve this problem, bacteria evolved channels which would sense salt by monitoring the electrical charge through an exchange of sodium and potassium. As history progressed, this channel took on new functions by sensing other ions like calcium and potassium. They later began to be used to fuel injections of compounds by pathogenic bacteria, and even later be used for motion. Even later in history, this group of proteins were used in nerve cells to send signals using electricity.
All cells are surrounded by a membrane which functions a lot like a soap bubble. These membranes are held together by soap-like molecules which have water-loving faces on the inside and outside of the cell. Their insides are repelled by water, and this dynamic is what keeps everything together. It’s somewhat pliable, and you can make the membrane harder or softer by adding a variety of different chemicals which insert themselves into that membrane.
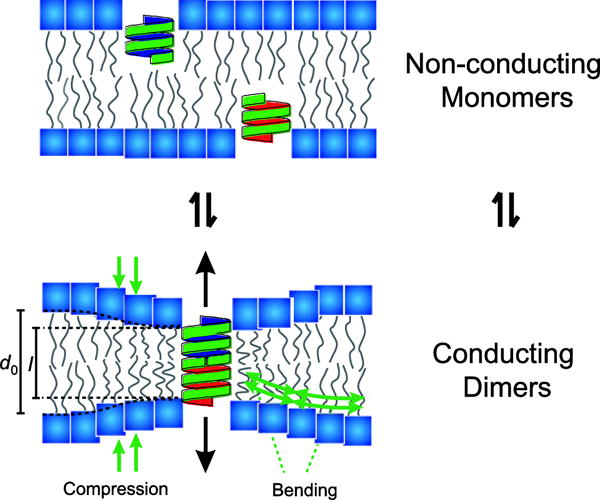
The best characterized electrical channel in bacteria is a sodium channel called NaChBac (I call it Nack-Back). Isoflurane inhibits this channel in bacteria, and similar ones in plants and animals. You can coax human cells to express this protein, clamp a very small glass tube on it, and then measure the change in voltage when you treat cells with isoflurane.
I know a lot of pop-culture science articles present proteins as biological machines, but that’s really not the case. To be frank, most of our proteins barely function. Proteins are, in essence, sticky strings which clump up in a way which just barely allows them to work. Get them too hot, too cold, or even just the wrong salt concentration, and they don’t work anymore. If you stick proteins in a membrane, they’re not really any different. If you fiddle with the characteristics of the membrane, that’s going to change the ability of the proteins in that membrane to function.
It was originally proposed by Ernst von Bibra that changes in the cell membrane could explain anesthesia. At the turn of the century, two scientists, Hans Meyer and Charles Overton independently published papers that demonstrated that the quality of the best anesthetics of the 1900s were correlated with their ability to dissolve in olive oil. For a long time, it was believed that anesthetics worked because they made the cell’s membrane too squishy or too hard for the ion channels to work efficiently.
If we time-jump forward a century or so, we now have the ability to measure how squishy cell membranes are. The antibiotic gramicidin has this cool ability to create channels in cell membranes. Simplistically, if a chemical makes the membrane more squishy, gramacidin will create a channel which will let things in* that destroys things we use to make cells shiny. Unfortunately, based on data from this assay, it doesn’t look like anesthetics affect the membranes enough to affect the activities of this protein under real-world concentrations.
So back to the drawing board…
So the Meyer-Overton theory was a great hypothesis, and it’s likely that the membrane interactions have some sort of effect on the sodium channels. It’s just not the main idea behind how they work.
We don’t actually have a good idea of how membrane proteins work in general. For awhile, it was thought that they formed rafts on the surface of the cell. However, more recent thinking seems to be that these “rafts” are an artifact of the methods we use to isolate these proteins. About a quarter of our proteins are the kind which are shoved into cell membranes, so we don’t know exactly what allows about 25% of our proteins function the way they do because they’re so hard to study.
A lot of the time, learning how these proteins work involve creating a protein crystal. We can’t predict the conditions under which a protein will crystallize. We have to subject it to a wide variety of conditions involving different salts, temperatures, solvents, and when all else fails, we shoot it into space. Because membrane proteins need to carry bits of the cell membrane with them, this presents an additional layer of difficulty above everything else.
If we want to say “anesthetics do this by binding to that“, we need to have a good idea of what we’re looking for before we start those experiments. So this is where bioinformatics come in. Some groups have started running massively complex computer simulations to determine how these compounds like isoflurane interact with these sodium channels.
But simulations are, well, simulations. Things which happen inside of a computer pretty frequently don’t interact with what happens in the real world.
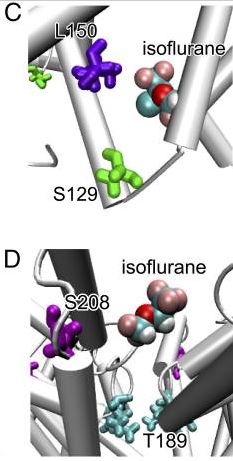
Since crystalizing proteins to take pictures is hard, there’s another thing we can do. We can slap fluorine molecules onto these channels, and use really strong magnets to take the pictures we need. This is called nuclear magnetic resonance.
So it turns out that isoflurane binds to the sodium channels at a place that’s just outside the cell. By binding here, it appears to affect the motion of the protein in a way which prevents it from efficiently being able to move atoms inside and outside of the cell.
Unfortunately, the technique here makes the proteins different from the ones found in the bacteria. These are also different from the ones used in mammals.
However, based on NMR data, it looks like isoflurane acts as an anesthetic by binding to the enzymes and affecting their ability to bend and efficiently transmit signals. It doesn’t look like they completely stop the protein from being able to transport ions, it looks like they stop them just enough to render you unconscious.
The Bottom Line
The reason why anesthetics work on plants, animals, and bacteria is because we all use a lot of the same processes to send signals. There’s only so many ways to send those signals, and the stuff plants and animals use to quickly send signals to distant regions of the body evolved early in evolutionary history. They worked pretty well, so everyone just kind of kept using the same proteins to do the same jobs even as the cells and bodies of these organisms went through drastic evolutionary changes.
Plants don’t have a reputation of moving very fast (or, like, at all), but there are a lot of plants which move very quickly. The most famous examples are of the venus flytrap closing their leaves in response to an insect, or Mimosa leaves closing in response to being touched.
There’s also some less famous examples, though. White mulberry flowers open at approximately half the speed of sound (mach 0.5), which is the quickest movement known to biologists. When you water plants, or burn them, they use electrical signaling to activate the necessary responses as quickly as possible**. Electrical signaling is as common in plants as it is in insects, and in bacteria, and is just as essential to their biology.
I should note that anesthetics like isoflurane need to affect many different nerve channels in similar ways, in a coordinated fashion which allows them to slightly shut things down. This is very different from an insecticide which completely and permanently shuts everything down at once. How and why anesthetics do this is still largely a mystery.
Another thing I need to point out is that there are a lot of different anesthetics and each of them has a different MOA. Each individual anesthetic also probably acts on several different systems at once. This post covers how one anesthetic interacts with one specific protein, in an experimental environment which might not actually reflect what’s going on in the human body. Hopefully the sheer complexity of the issue comes through.
Either way, though, using anesthetics on plants and bacteria has demonstrated that their modes of action are largely universal. We need to know how these things work in humans, and knowing that their modes of action were universal was a monumental first step in solving that mystery.
For my dissertation, I was initially very interested to see whether this sort of quick signaling was involved in the type of shade avoidance response I’m currently studying. Unfortunately, because anesthesia acts on so many systems all at once, I decided that other probes would likely be more effective once we get a few other experiments out of the way first.
It’s still an idea I’m planning on pitching, but I’d like to find more specific blockers which might work before I do.
Works Cited
Charalambous, K., & Wallace, B. A. (2011). NaChBac: the long lost sodium channel ancestor. Biochemistry, 50(32), 6742-6752.
Choi, W. G., Hilleary, R., Swanson, S. J., Kim, S. H., & Gilroy, S. (2016). Rapid, long-distance electrical and calcium signaling in plants. Annual Review of Plant Biology, 67, 287-307.
Herold, K. F., Andersen, O. S., & Hemmings, H. C. (2017). Divergent effects of anesthetics on lipid bilayer properties and sodium channel function. European Biophysics Journal, 46(7), 617-626.
Kinde, M. N., Bondarenko, V., Granata, D., Bu, W., Grasty, K. C., Loll, P. J., … & Xu, Y. (2016). Fluorine-19 NMR and computational quantification of isoflurane binding to the voltage-gated sodium channel NaChBac. Proceedings of the National Academy of Sciences, 113(48), 13762-13767.
Ouyang, W., Jih, T. Y., Zhang, T. T., Correa, A. M., & Hemmings, H. C. (2007). Isoflurane inhibits NaChBac, a prokaryotic voltage-gated sodium channel. Journal of Pharmacology and Experimental Therapeutics, 322(3), 1076-1083.
Footnotes
*I’m skipping over a lot of hardcore biochemistry, and math that’s even more hard-core. Basically, you put a fluorophore called ANTS into a cell, and gramicidin allows a thallium ion into the cell which stops ANTS from glowing. You then compare the brightness of cell cultures to one another to get a measurement of the ability of gramicidin to create pores. This ability is directly related to the squishieness or hardness of the cell membrane. It’s really cool, and while I love to geek out over stuff like this, I’m trying to keep things as simple as I can. Here’s a more in-depth description for anyone who wants to know more.
**Yes, this is similar to how nerves work in animals. The presence of this kind of signaling does not say anything about sentience in plants. Without getting into too much of a debate on this, it’s widely accepted that plants are not sentient. There are articles in the peer-reviewed literature which make this claim, but unfortunately Bad Science can slip through the review process in a number of ways and these articles do not stand up to scrutiny. We’ve written about one similar case before.


You must be logged in to post a comment.