Written by Joe Ballenger
In December 2019, the world changed forever. I know that’s a bit of a heady way to begin this article, but it’s true. The current SARS-CoV-2 pandemic has changed a lot of the way people live, and exposed a lot of societal inequalities in the US. Over a quarter-million people have died, and in The West (where Joe is currently living), more than a handful of states have had 1 in every 1000 citizens or more die from the disease.
With the announcement of several promising vaccine candidates, I do think there’s hope on the horizon…which is why I waited until Thanksgiving week to write this. There’s a good reason to be thankful to insects, and in a weird way, we can be thankful for the field of insect pathology for making some of these candidates a possibility.
Baculoviruses are kind of famous for making “zombie caterpillars”. The way they kill insects is admittedly pretty gruesome. The caterpillar eats the virus, and when the infection is almost ready to kill the insect, the caterpillar crawls up to the highest point it can find. The virus then releases enzymes which kill the caterpillar and almost completely dissolve it, and rain infectious particles down on the forest below to start the cycle over.
It’s definitely a bit shocking, but the viruses are harmless to people. For that reason, they’re really important biological control agents in a lot of agricultural systems.
While they’re important for agriculture, they’re arguably more important in the field of medicine. These viruses have been a boon to the field of biochemistry, because they’re really important tools we can use to make proteins…like the SARS-CoV-2 spike protein.
Why Baculoviruses?
There’s a number of ways to make vaccines, from using bits of viral protein to weakened viruses, viral vectors. Stuff like that.
The vaccine candidates which are the furthest along from Moderna and Pfizer, are mRNA vaccines which teach your body to recognize the SARS-CoV-2 genetic material. They’re clearly effective, but they are based on mRNA, which is a notoriously unstable material. They need to be stored at temperatures colder than a home freezer, which could be an issue when distributing in rural areas. Proteins don’t have this problem, so there’s use for multiple types of vaccines.
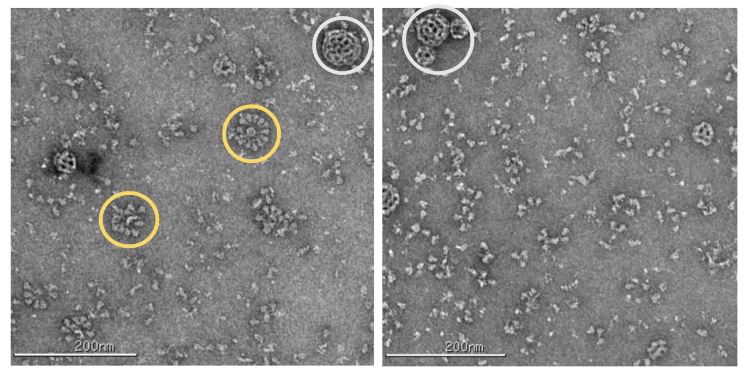
The Novavax vaccine uses the protein SARS-CoV-2 to enter cells. They express this protein in insect cells, and the spike proteins form little balls while in solution (yellow circles) with detergent. They also inject some micro-balls (white circle) which jump-start the immune response.
The important thing here is that these are not active viruses. They don’t contain any genetic material, so they can’t reproduce or cause disease. I’d be happy to do another post on the biochemistry of the vaccine candidates if there’s enough interest, but since I want to focus more on the baculovirus part…this is as far as I’m going to get into that topic.
So why are insect cells used to make proteins?
The cell line used by Novavax to make their proteins are SF9 cells, originally isolated from the ovaries of pupal Spodoptera frugiperda. Fall Armyworm is a pretty intense pest. It’s one of the few insects which is a billion-dollar pest in agriculture, and it’s recently been established in Africa. The interest in controlling this insect has made it a workhorse for molecular biology, and it’s one of the major insect models. This interest is what led to the creation of the SF9 cell line.
There’s a number of cell lines which can be used to make proteins, but there’s trade-offs with each different cell line. Chinese Hamster Ovary (CHO) cells are typically used, and have a pretty high protein yield; ~7g of protein per liter of culture. SF9 cells are a bit lower, around the neighborhood of 1 gram of protein per liter of culture.
Beyond yield, there’s a few other things to consider. Cost effectiveness is one, but I wasn’t able to find numbers for CHO cells. SF9 can cost between $2,000 to $5000 for a gram of protein. This sounds like a lot of money, and it is, but a gram of protein is a lot of protein. The Armour Hot Dog company purified a kilogram of RNAase A (a type of RNA cutting protein), and this relatively small amount lasted the entire scientific community decades.
Another consideration of these expression systems is what biochemical pathways are present in the cells. With mammal cells, there’s always the possibility that they’re too similar to your host because they’ll make stuff which interfere with your proteins. They might also slap different sugar groups on the proteins, which can be either good or bad. If they leave the sugars off, that might make the proteins more avaliable to make antibodies in vaccines. Sometimes, the cells make sugars which are more similar to natural proteins than mammal cell lines.
So…it really depends on a lot of different factors. Some of this stuff might be considered trade secrets, stuff companies hold back to make themselves a bit more competitive. It makes science writing hard, but these companies also need to be able to compete with one another.
As a side note, all of the companies involved in COVID vaccine production have pledged to sell the vaccines at cost for the duration of the pandemic (although I haven’t seen any define an endpoint). I don’t think this will be a problem in the forseeable future.
How do baculovirus expression systems work?
Baculoviruses, specifically, are really good for protein production. After the virus is done with the cells, more than half the protein in the cell is viral protein. So if you toss the protein you want to express behind one of the highly expressed viral proteins in the cell, you can get a really good protein yield.
More or less, they insert the gene of interest into a baculovirus genome that’s then amplified in E. coli. After they isolate the baculovirus genome, they then use a mild detergent to get the virus genome into the cells. Once the viral genome is inside the insect cells, it makes viruses and your protein of interest.

It’s a pretty complicated procedure, but there’s been a lot of advances over the years. The viruses themselves are even modified in a variety of ways to make protein expression easier, more efficient, and better match what you need them to do. It’s an interesting story, perhaps worth a post if folks are interested, but this is a general backbone of what’s done.
Conflict of Interest and Bottom Line
Whenever I write about biotechnology I do have to disclose that I worked for a pharmaceutical company, on the agricultural side of things. Specifically, I worked as a permatemp…an incredibly exploitative and problematic form of labor which many biotech companies use to hire a large proportion of their scientists without paying them benefits. So I need to disclose a potential COI, and mention that I’ve got a complicated stance about how these companies operate.
So that being said, with those cards on the table, this is a vaccine which will ultimately save hundreds of thousands of lives in the US alone…and millions across the world. I’ll be getting it as soon as I can, and encouraging everyone in my social circle to do the same. The only way we’re gonna get out of this is by working together to help our communities, and getting the COVID-19 vaccine is just a part of it.
We also need to talk to our friends and families into getting it, as well.
Works Cited
S. Bangaruet al., Science10.1126/science.abe1502 (2020).
Chambers, A. C., Aksular, M., Graves, L. P., Irons, S. L., Possee, R. D., & King, L. A. (2018). Overview of the baculovirus expression system. Current protocols in protein science, 91(1), 5-4.
Jarvis, D. L. (2009). Baculovirus–insect cell expression systems. In Methods in enzymology (Vol. 463, pp. 191-222). Academic Press


You must be logged in to post a comment.